
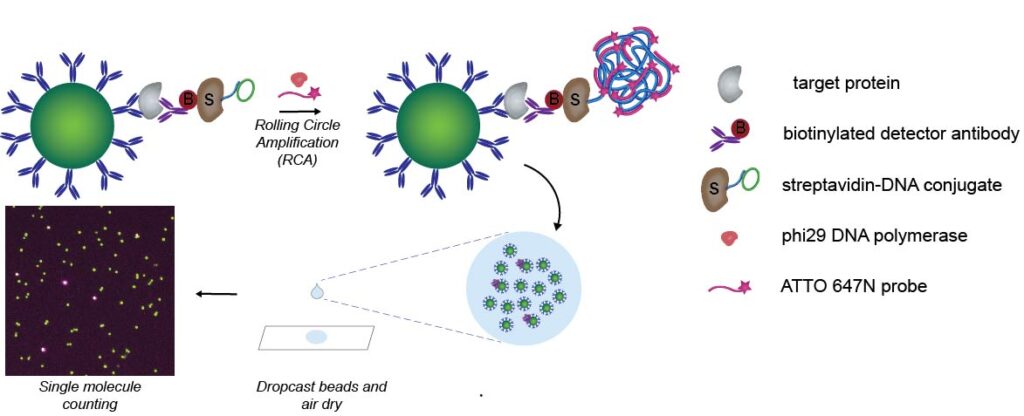
Ultrasensitive Detection
The Simoa technology enables 100- to 1000-fold improvements in sensitivity over conventional enzyme-linked immunosorbent assay (ELISA) techniques, thus opening the window to measuring proteins at concentrations that have never been detected before in various biological fluids such as plasma or saliva. However, there remain many potential disease-associated biomarkers that exist in these biofluids at concentrations below the current detection limit of Simoa. We are therefore developing improved methods to further push the boundary of sensitivity to detect even lower biomolecule concentrations, using on-bead signal generation strategies as well as droplet-based digital ELISA platforms.
Point-of-Care Diagnostics
Much of our research is driven by pressing clinical needs. For our assays to reach the clinic and resource-limited settings, they need to be built on new platforms that are smaller, cheaper, and faster, without sacrificing their single-molecule sensitivity. We are developing new technologies to carry out our tests—from sample processing to liquid handling to signal readout—that are automated, simple, and robust. This work synthesizes our members’ and collaborators’ expertise in chemical engineering, microfluidics, applied math, and optics.
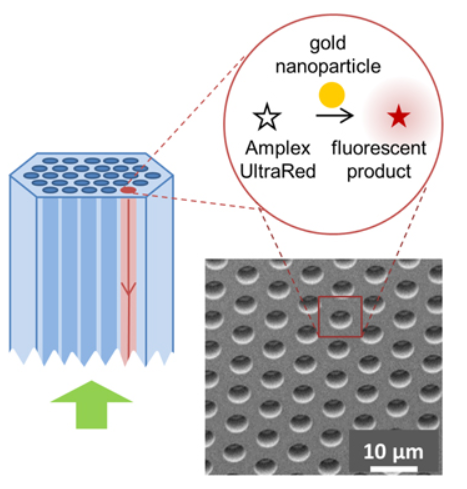
Catalytic Activity of Single Nanoparticles
Metal nanoparticles are important catalysts in a variety of industrial applications. However, their catalytic properties are not fully understood, as they are highly dependent on the details of particle structure, including shape, size, composition, and crystallinity. Using a method analogous to that described for single molecule enzyme activities, we can explore the individual catalytic activities of a heterogeneous ensemble of nanoparticles. The catalytic activities of hundreds of individual nanoparticles, as well as the detailed kinetics of the reactions, can be measured and statistically analyzed. These catalytic properties will be correlated with the optical and structural properties of the nanoparticles, via optical spectroscopy and scanning electron microscopy, respectively.