Alpha-synuclein species in extracellular vesicles
Pathological alpha-synuclein isoforms play a significant role in PD pathophysiology. The pathological differences observed in the brain tissue of PD patients compared to neurological controls (NCs) motivated researchers to explore alpha-synuclein as a biomarker for PD. However, multiple studies have demonstrated that the measurement of alpha-synuclein species circulating in CSF and plasma does not yield high diagnostic accuracy. Extracellular vesicles (EVs) secreted by neurons should provide a more accurate reflection of cellular conditions and should preserve alpha-synuclein species in their original form. The goal of this project is to evaluate alpha-synuclein species internal to EVs derived from CSF and plasma as biomarkers for PD. We aim to develop a pipeline to determine the concentrations of total alpha-synuclein, pSer129 alpha-synuclein, and aggregated alpha-synuclein inside EVs with high sensitivity and accuracy. We will then apply our methods to profile alpha-synuclein isoforms internal to EVs in CSF and plasma samples collected from a well-characterized cohort of PD patients and NCs.
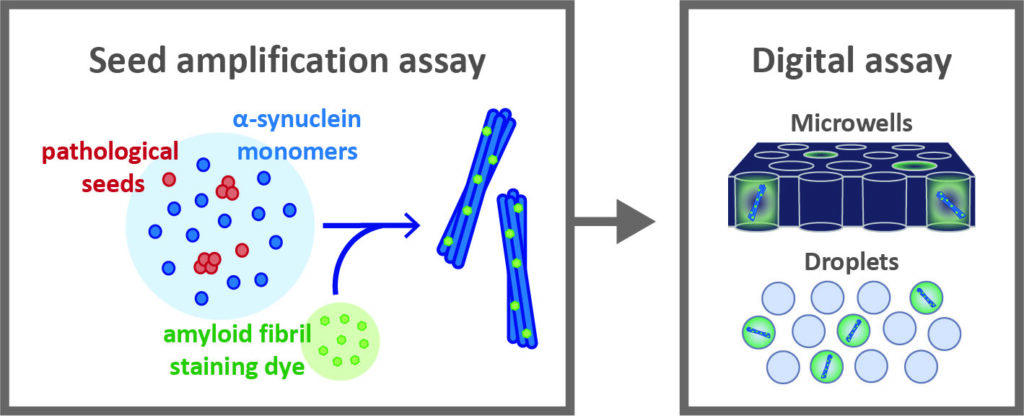
Digital seed amplification assays
Currently, PD is a clinical diagnosis. Physicians identify the disease primarily based on symptoms such as cogwheel rigidity, bradykinesia, and tremors. Despite the availability of clinical guidelines, under-diagnosis and misdiagnosis are not uncommon. Therefore, one of the major intermediate goals for curing PD is finding effective methods for diagnosing the disease, ideally before symptom onset, allowing clinicians to track disease progression and implement preventative treatment strategies. Alpha-synuclein aggregates represent a promising biomarker for PD, as their intraneuronal accumulation strongly correlates with disease severity. Pathological forms of alpha-synuclein can be detected in the biofluids of PD patients using seed amplification assays (SAAs); however, in their present format these assays are not quantitative and suffer from a lack of reproducibility.Our overarching goal is to develop a digital alpha-synuclein SAA to measure the concentration of pathological aggregates in patient biofluids. We aim to establish a robust digital SAA to measure the number of pathological aggregates present in cerebral spinal fluid (CSF) samples. In the future, we aim to optimize the assay for detecting alpha-synuclein aggregates in plasma-derived extracellular vesicles, bypassing the need for an invasive spinal tap and paving the way for early PD screening. A quantitative SAA has the potential to yield new insights into the evolution of PD and could be used as a companion diagnostic in clinical trials targeting ⍺-synuclein aggregation.
Funding: The Michael J. Fox Foundation