Alzheimer’s Disease
Immuno-dementia biomarker discovery using extracellular vesicle liquid biopsy
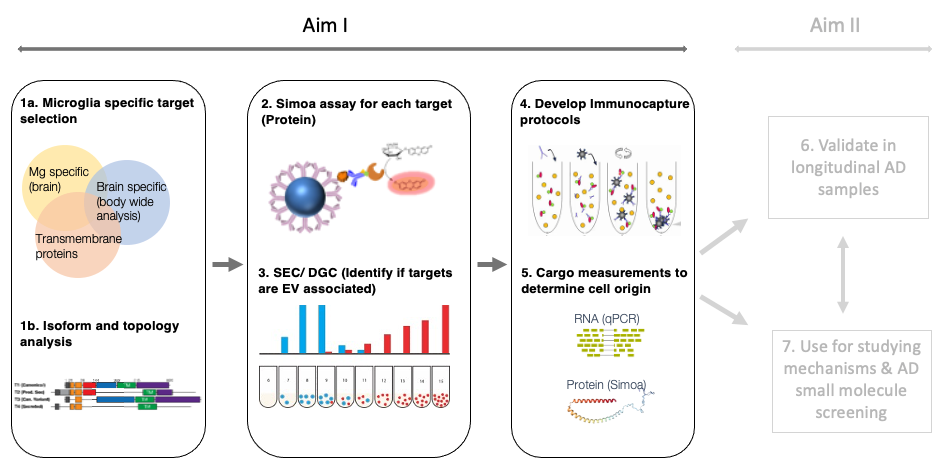
Microglia are important for maintaining the health of the central nervous system. Aberrant microglial response to amyloid pathology has been implicated as one of the key factors in Alzheimer’s disease progression. Microglia have several roles in the brain and can exhibit a few unique gene signatures depending on the role in which they are engaged. Gene signatures are traditionally measured from fresh tissue samples, such as biopsies, which is not a feasible sampling method for neurological disease researchers. Extracellular vesicles (EVs) are lipid bilayer particles secreted by all cell types that encapsulate proteins, RNAs, and metabolites. EVs provide a snapshot of the cells from which they are derived and can provide a signature of cells at the time of release. The goal of this project is to develop protocols for microglial EV enrichment and analysis and to use these protocols to assess the efficacy of novel therapeutics in ongoing Alzheimer’s disease clinical trials.
Funding: The Genetics Guided Dementia Discovery Platform of Eisai Inc . and Open Philanthropy/Good Ventures